|
Clearance is reduced in patients with renal impairment, but no dose adjustment is currently recommended by the manufacturer.Ī prospective cohort study is investigating patients taking dabigatran who present with life-threatening bleeding or require surgery that cannot be delayed. It is probably catabolised with 32% of the dose being excreted in the urine within six hours of infusion. 1 There was a rapid improvement in clotting studies such as thrombin time and activated partial thromboplastin time. 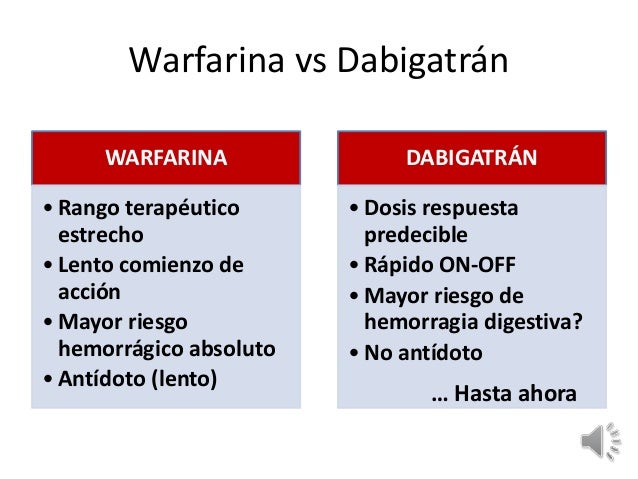
After idarucizumab doses of 2 g or more, they remained close to the lower limit of quantification during 72 hours of observation. Idarucizumab immediately bound to dabigatran so unbound dabigatran concentrations fell quickly. They were given dabigatran for a few days then, within two hours of the last dose, they were infused with idarucizumab or a placebo. To test the concept that idarucizumab would reverse the effect of dabigatran a trial was carried out in 47 healthy men. The affinity of this antibody for dabigatran is greater than the affinity of dabigatran for thrombin. The development of idarucizumab involved genetically engineering a humanised monoclonal antibody fragment.  
Idarucizumab has been developed to reverse the effect of dabigatran, a direct thrombin inhibitor. Reversal of anticoagulation may be required if the patient develops severe bleeding or requires emergency surgery. Australian Medicines Handbook section 7.4Ī limiting factor in the use of the newer oral anticoagulants is that, unlike warfarin, there have been no antidotes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
